On October 31, 2024, SIGNDO Biotech (Suzhou) Co., Ltd. (hereinafter referred to as "SIGNDO Biotech") announced that its independently developed DGPR1008 injection, a near-infrared tumor-targeted fluorescent contrast agent with independent intellectual property rights, has completed the enrollment of the first patient in the Phase II clinical study at the leading hospital, the Affiliated Hospital of Qingdao University, and the corresponding surgical operation has also been accomplished.
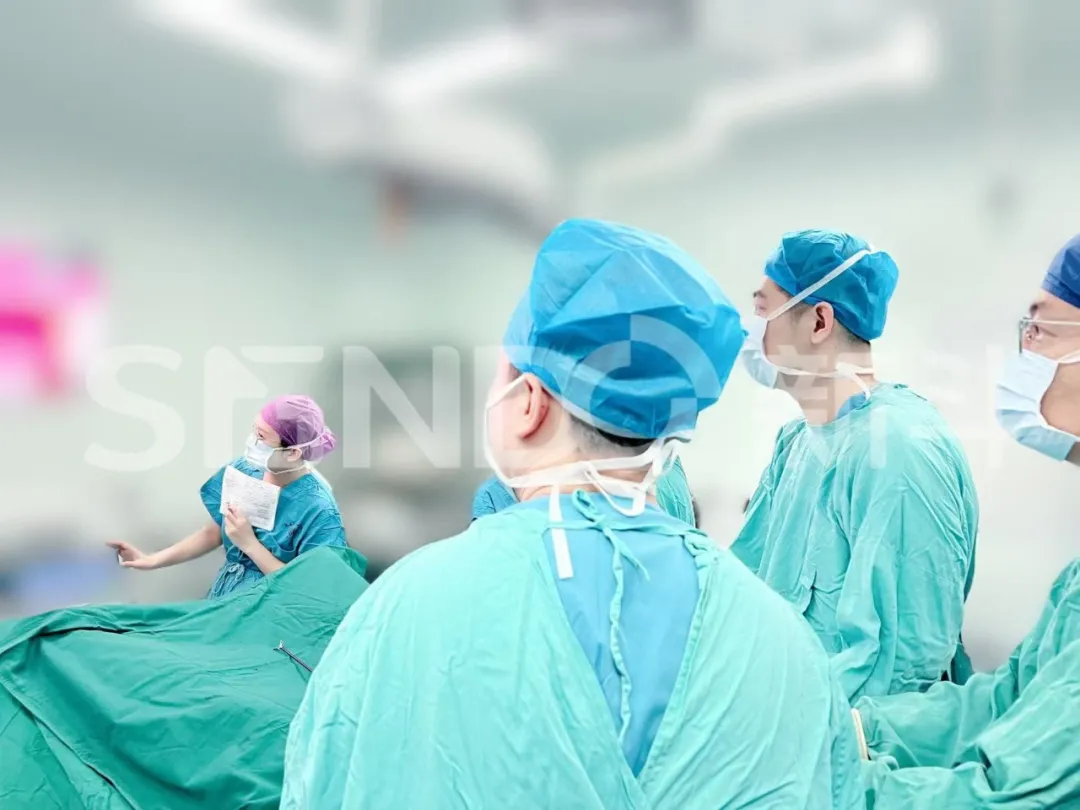
The aim of this study is to investigate the safety profile and the intraoperative imaging efficacy of DGPR1008 injection in patients with prostate cancer. Previously, the company had successfully concluded the Phase I clinical trial regarding the safety, tolerability, and pharmacokinetic parameters of a single administration of DGPR1008 injection in healthy volunteers. The results demonstrated that it exhibited favorable safety and tolerability characteristics in healthy individuals, thereby providing robust support for the Phase II clinical study.
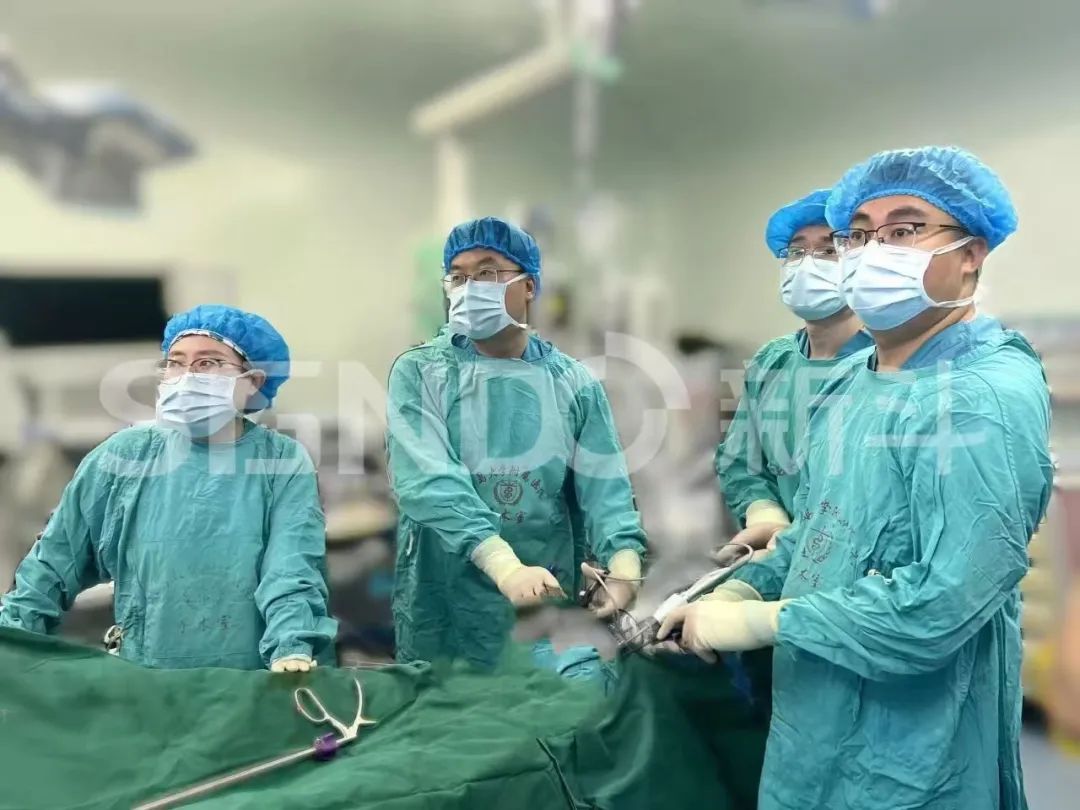
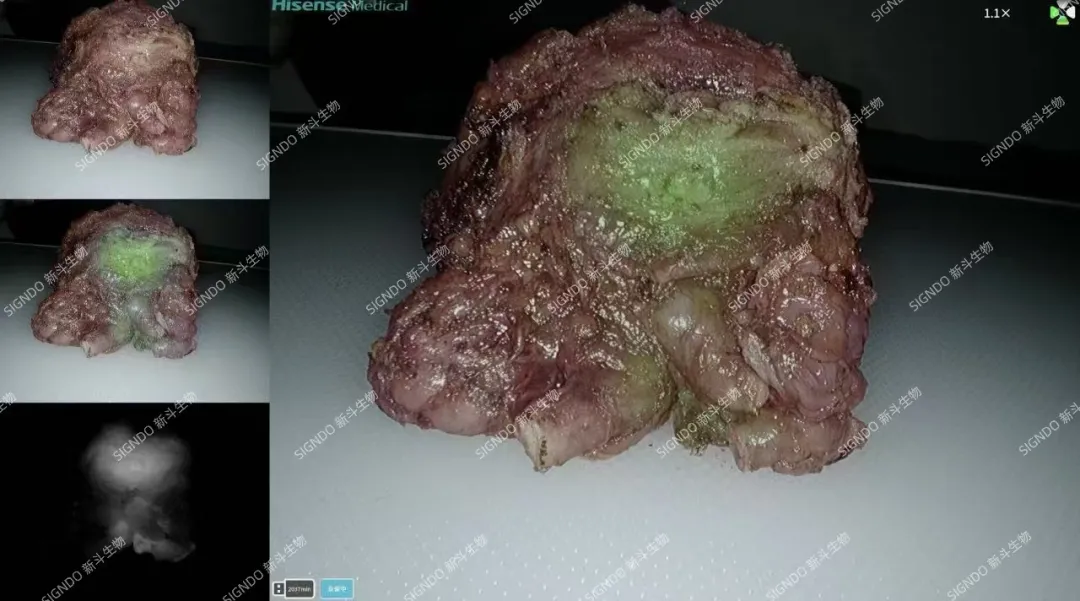
Haitao Niu, a member of the Standing Committee of the Party Committee and vice president of Qingdao University, along with Yu Cao, the director of the Office of the Clinical Trial Center and the Phase I Clinical Research Center of the Affiliated Hospital of Qingdao University, acting as the principal investigator (PI),in conjunction with the teams led by Director Xuecheng Yang and Director Yanjiang Li from the Department of Urology of the Affiliated Hospital of Qingdao University, stated: Currently, there is no targeted fluorescent contrast agent product available in the global market for prostate cancer. As the first near-infrared tumor-targeted fluorescent contrast agent approved for clinical trials in China, DGPR1008 injection has completed its Phase I study, showing good safety and tolerability. It can precisely image during prostate cancer surgeries, possess excellent tumor-targeting capabilities for prostate cancer, and assist surgeons in accurately localizing the tumor and visualizing its margins, thereby minimizing iatrogenic damage to normal tissues. We look forward to accelerating the clinical research process of DGPR1008 injection to promptly introduce this innovative drug to a large number of prostate cancer patients.

Jing Zhao, the founder and CEO of SIGNDO Biotech, remarked: DGPR1008 injection
is the pioneer near-infrared tumor-targeted fluorescent contrast agent approved
for clinical trials in China. Based on the excellent safety manifested by our
preclinical and Phase I trial data, we promptly initiated this study with the
intention of swiftly bringing this innovative drug to the benefit of a large
number of prostate cancer patients. The seamless accomplishment of the
administration to the first patient and the surgical procedure represents a
significant milestone in the clinical development pathway of DGPR1008
injection. We will be even more determined in continuously and expeditiously
promoting the clinical development process of DGPR1008 injection and the research
and development of other pipelines, thereby making greater contributions to the
innovative R&D and industrial progress of targeted fluorescent contrast
agents in China.