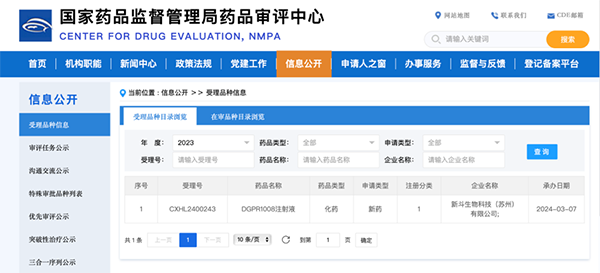
On March 7, 2024, according to public information from the Center for Drug Evaluation (CDE) of the National Medical Products Administration, the Investigational New Drug (IND) application for "DGPR1008 Injection" submitted by SIGNDO Biotechnology (Suzhou) Co., Ltd. (hereinafter referred to as "SIGNDO Biotech") was officially accepted (Acceptance Number: CXHL2400243). This marks the first-in-class PSMA-targeted fluorescent contrast agent product to be officially accepted in China. The acceptance by the CDE is expected to further advance the clinical translation and development of tumor-targeted fluorescent contrast agents. Meanwhile, SIGNDO Biotech is actively progressing its pipelines of tumor-targeted fluorescent contrast agents for other indications.
01 Milestone Event
DGPR1008 Injection is the first-in-class tumor-targeted fluorescent contrast agent developed by SIGNDO Biotech. In 2023, an investigator-initiated trial (IIT) was conducted in grade A tertiary hospital, during which no drug-related adverse events or serious adverse events occurred, and the clinical benefits were highly significant.
02 Product
DGPR1008 Injection (Tumor-targeted Fluorescent Contrast Agent), independently developed by SIGNDO Biotech, is a novel drug designed for real-time targeted fluorescent imaging in prostate cancer. Administered intravenously before surgery, it targets tumor cells to enable real-time intraoperative fluorescent imaging. This assists doctors in accurately determining positive surgical margins for the complete resection of tumor lesions, significantly reducing damage to normal tissues and aiding in the identification of small tumor lesions and metastatic lymph nodes.
03 Classic case
Male patient, 68 years old
The patient was admitted to the hospital one week after a routine physical examination revealed elevated PSA levels.
Upon initial presentation, the patient's PSA was >100 ng/ml.
Magnetic Resonance Imaging (MRI): Prostate cancer was detected in the central gland and peripheral zone on the left side of the prostate with enlarged lymph nodes in the left pelvic region.
Biopsy Pathology: Prostate cancer with a Gleason score of 4+4=8, involving both sides. The patient had significantly elevated PSA levels and enlarged pelvic lymph nodes, indicating a high tumor burden and poor prognosis.
To achieve complete tumor resection and improve long-term survival, the patient was administered DGPR1008 Injection before surgery. Post-administration, the patient underwent a fluorescence-guided laparoscopic radical prostatectomy with pelvic lymph node dissection.
DGPR1008 Injection successfully targeted the tumor lesions and metastatic lymph nodes. When the fluorescence mode was activated, the prostate cancer lesions, metastatic lymph nodes, and other tumor-infiltrated tissues fluoresced green due to the binding of the tumor-targeted fluorescent contrast agent. After completing the prostatectomy according to the standard surgical procedure, the fluorescence mode was reactivated to inspect the surgical field. Residual fluorescent tissue was observed at the apex of the tumor bed, suggesting residual tumor tissue. The residual fluorescent tissue was excised under fluorescence guidance, and pathological examination confirmed the presence of malignant tissue.
In summary, DGPR1008 Injection has demonstrated significant clinical value in intraoperative tumor lesion visualization, positive margin determination, and metastatic lymph node localization.
Focus on Tumor-targeted Fluorescent Contrast Agent
SIGNDO Biotech was founded in Suzhou in February 2021, focusing on the R&D and industrialization of tumor-targeted fluorescent contrast agents. With the goal of "Illuminate Cancer, Precision Surgery," SIGNDO Biotech has established a globally leading RT TFC® technology platform to build a multi-cancer product portfolio, aiming to become a pioneer in the field of targeted fluorescent contrast agents in China.
TFC® technology platform to build a multi-cancer product portfolio, aiming to become a pioneer in the field of targeted fluorescent contrast agents in China.