SIGNDO Biotech announced that the first-in- class batch of subjects has been enrolled and dosed in the Phase I clinical trial of DGPR1008 Injection, the first-in-class near-infrared tumor-targeted fluorescent contrast agent product to receive clinical approval in China. The trial is being conducted at The Affiliated Hospital of Qingdao University.

Following the clinical approval on May 20, SIGNDO Biotech rapidly advanced the Phase I clinical trial, completing the initial dosing on June 24. The dosing process went smoothly, and post-dosing observation has been completed. The subjects demonstrated good tolerance during the observation period and were successfully discharged on June 27.
1. Restoring Hope: A New Treatment Approach for Prostate Cancer Patients
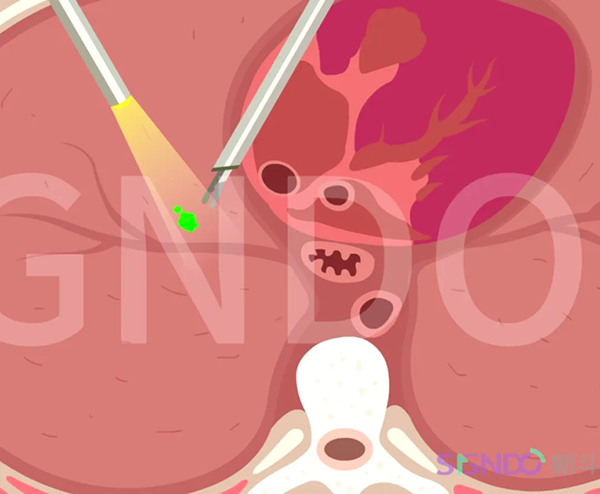
Prostate cancer is one of the most common tumors in the male reproductive system. According to the 2020 WHO Global Cancer Statistics report, the incidence and mortality rates of prostate cancer in China rank fourth and ninth among all malignant tumors respectively, following breast cancer, lung cancer, and colorectal cancer. In recent years, the incidence and mortality rates of prostate cancer have been rapidly increasing. Surgical resection remains the preferred treatment for most primary cancers. In radical prostatectomy, an overly extensive resection may damage normal tissues, affecting functions such as urinary control, while an insufficient resection may result in positive surgical margins, leading to a high risk of recurrence. During pelvic lymph node dissection, it is also challenging for surgeons to distinguish between metastatic and non-metastatic lymph nodes in real-time during surgery.
The advent of near-infrared targeted fluorescent imaging agents has significantly assisted physicians, utilizing a novel technology that allows real-time visualization of the tumor's location, size, boundaries, and metastatic lymph nodes during surgery. This technology also helps reduce the likelihood of tumor metastasis or recurrence in patients. There has been a gap in this field in China. The completion of the initial enrollment in the Phase I clinical trial of DGPR1008 Injection marks a significant step forward in bringing this innovative product closer to benefiting a broader global patient population.
2. About DGPR1008 Injection
DGPR1008 Injection:
DGPR1008 Injection is a near-infrared tumor-targeted fluorescent contrast agent product independently developed by SIGNDO Biotech, possessing independent intellectual property rights. Administered intravenously before surgery, it enables precise intraoperative imaging by illuminating tumor cells in real-time. The product offers advantages such as "high sensitivity, strong specificity, and a favorable tumor-to-background ratio (TBR)," which assists surgeons in clearly locating the tumor, visualizing its boundaries, and identify tumor margins. This significantly reduces damage to normal tissues and also aids in identifying occult tumor lesions and metastatic lymph nodes.
3. About SIGNDO Biotech
SIGNDO Biotechnology is an innovative biotechnology company specializing in the R&D, as well as the industrialization of tumor-targeted fluorescent contrast agents and related products. Since SIGNDO Biotech founding in 2021, our team has been dedicating to providing doctors with tools required for tumor visualization. Goal of "Illuminate cancer, Precision surgery ", SIGNDO Biotech committed to help surgeons achieve a more complete surgical resection and help improve patient outcomes. Real-time Targeted Fluorescent Conjugate(RT TFC®)technology platform is a leading global technology platform, which can utilize small molecule fluorescence conjugate technology to build a multi-cancer product portfolio for intraoperative visualization of tumors by surgeons.
TFC®)technology platform is a leading global technology platform, which can utilize small molecule fluorescence conjugate technology to build a multi-cancer product portfolio for intraoperative visualization of tumors by surgeons.